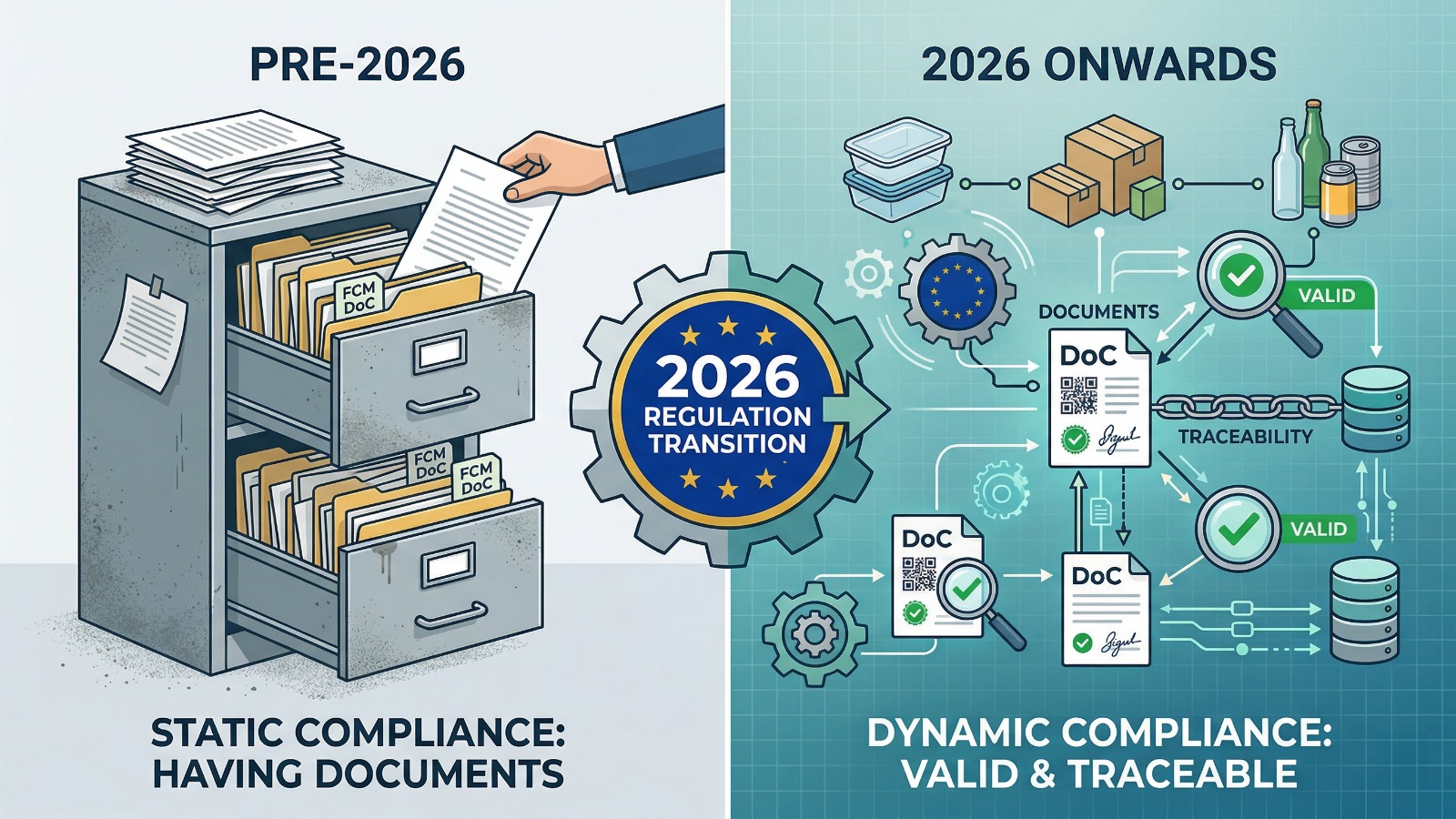
### A declaration alone is no longer enough<br />For many packaging teams, compliance used to mean having a declaration of conformity somewhere in the file. In 2026, that expectation becomes narrower because customers, importers and market checks increasingly focus on whether the declaration is current, product-specific and linked to usable supporting records.
### Traceability gives the document practical value<br />A declaration is far more useful when it can be connected to batches, product codes and supply records that identify the exact packaging item in circulation. Without that traceability layer, the file may exist formally but still fail to answer the questions that arise during audits or complaint handling.
### Version control matters when regulations and product details move<br />Companies often keep documents for years, but that habit becomes harder to defend when formulations, suppliers or regulatory expectations change. Packaging teams need to know which version applies, when it was updated and whether the supporting information still matches the product being sold now.
### Intended use should be stated clearly, not assumed<br />Temperature range, contact conditions, food type and known limitations all affect how a declaration should be read. A document is weaker when the use case is left broad or implied, because customers cannot easily tell whether the packaging file actually fits their application.
### Supplier records need to stay connected to the same item<br />Technical sheets, declarations and related reports help only when they describe the same packaging structure and commercial article. If the file is assembled from loosely related documents, later verification becomes slower and much less convincing.
### Better compliance comes from maintaining the document chain<br />In practice, 2026 pushes packaging teams toward active document management rather than passive storage. When declarations, batch linkage, intended use and supporting records remain aligned, the packaging file becomes easier to update, explain and defend over time.